My reelmaking is quite dependent on the anodizing process. Running this process consistently means controlling three conditions:
1. Current density (12 A/in^2)
2. Temperature (68 deg F)
3. Acid concentration
Recently it occurred to me that my acid mixing is haphazard. My reference book discusses acid concentration in terms of “percent by volume”, and I now realize that I do not know what this means. The problem is that 10 fluid ounces of water mixed with 10 fluid ounces of acid yields a solution that is less that 20 fluid ounces volume. When I mix the water and acid, I first put a measured volume of distilled water in a container, and then add enough automotive battery acid (already an acid-water mixture) to make up twice the volume. I am not sure how much acid this takes.
To monitor acid concentration on an ongoing basis, one needs a hydrometer (any auto parts store).

I bought this one because it shows specific gravity, not just some colored balls floating.
Whatever concentration I arrived at, I am getting satisfactory results (although I still am not capable of measuring film thickness) and I intend to maintain this concentration. My acid solution measured SG = 1.15 . The container of my source battery acid says SG = 1.265 .
A table of sulfuric acid concentration (on a percent by weight basis) vs specific gravity can be found here. Below, I have plotted the data.
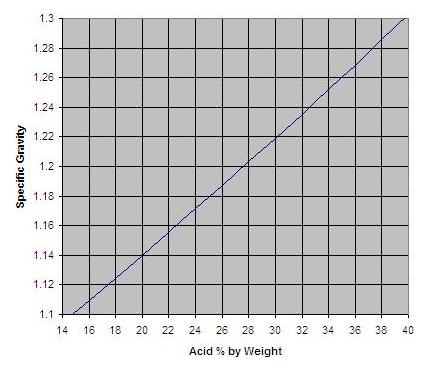
So I am using a solution that is about 21% acid by weight.

This was really helpful, thanks!